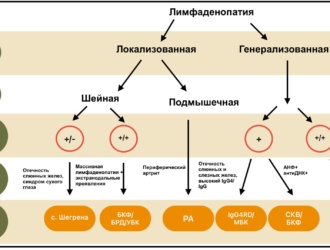
OBJECTIVE:
OBJECTIVE:
Febuxostat has superior renal safety to allopurinol, but data on its hepatic safety are limited. Thus we compared the hepatotoxicity of febuxostat and allopurinol, and the clinical factors associated with hepatotoxicity, in patients with gout and fatty liver disease (FLD).
METHODS:
We included gout patients treated with allopurinol or febuxostat who were diagnosed with fatty liver based on ultrasonography or computed tomography. Hepatotoxicity was defined as follows: (1) elevation of aspartate aminotransferase (AST)/alanine aminotransferase (ALT) at least 3x the upper limit of normal, when the baseline AST/ALT was normal; or (2) doubling of the baseline AST/ALT, when the baseline AST/ALT was elevated. The factors associated with hepatotoxicity were evaluated by Cox regression analysis.
RESULTS:
Of 134 patients identified with gout and FLD, 32 (23.9%) received febuxostat and 102 (76.1%) received allopurinol. There were no significant differences in age, body mass index, comorbidity, or disease severity between the groups; however, the incidence of hepatotoxicity was significantly lower in the febuxostat group (3/32, 9.4%) than in the allopurinol group (36/102, 35.3%, p = 0.005). Diabetes (HR 3.549, 95% CI 1.374−9.165, p = 0.009) and colchicine use (HR 11.518, 95% CI 5.515−24.054, p < 0.001) were associated with a higher risk of hepatotoxicity, whereas febuxostat use was associated with a lower risk of hepatotoxicity (HR 0.282, 95% CI 0.086−0.926, p = 0.037).
CONCLUSION:
In the 32 patients studied, febuxostatwas well tolerated in patients with gout and FLD. However, the presence of diabetes and colchicine use may increase the risk of hepatotoxicity.
J Rheumatol. 2019 May; 46 (5): 527 — 531
Lee JS, Won J, Kwon OC, Lee SS, Oh JS, Kim YG, Lee CK1, Yoo B, Hong S.
doi: 10.3899/jrheum.180 761